Isotopic Dilution for PFAS: The Gold Standard for Accurate Environmental Analysis
What isotopic dilution is and why it delivers more accurate and defensible PFAS measurements.

Isotopic dilution is widely recognized as the gold standard for PFAS testing. While many environmental professionals know it represents the highest level of analytical quality, few understand why.
Table of contents:
What Is Isotopic Dilution?
Isotopic dilution is a quantitative analytical technique where a known amount of an isotopically labeled version of a compound is added to a sample before analysis. For PFAS testing, this means adding compounds like ¹³C-labeled PFAS standards to water, soil, or tissue samples before the sample is extracted.
In short: Isotopic dilution is a method that corrects for losses and matrix effects by measuring a native compound relative to a chemically identical, isotopically labeled internal standard.
Why PFAS Analysis Needs Isotopic Dilution
Per- and polyfluoroalkyl substances (PFAS) are:
- Tested at ultra-trace levels (ng/L or lower)
- Prone to matrix interference (PFAS are sensitive to biologics, metals, sediment, et.)
- Easily lost during sample prep (PFAS adsorb to many surfaces such as plastic or glass)
- Found in complex environmental matrices (groundwater, wastewater, tissues, sludges, etc.)
Because PFAS methods often rely on solid phase extraction (SPE) and LC-MS/MS, any loss during extraction or matrix interference variability directly impacts accuracy.
Isotopic dilution corrects for:
- Extraction recovery losses
- Matrix ion suppression/enhancement
- Instrument variability
What is an Isotope?
Isotopes are atoms of the same element that have the same number of protons but different numbers of neutrons. Because they have different neutron counts, isotopes have different atomic masses, but they share almost identical chemical properties.
In nature, 99% of carbon atoms are ¹²C, meaning they have 6 protons and 6 neutrons. The other carbon isotopes ¹³C (6 protons and 7 neutrons) and ¹⁴C (6 protons and 8 neutrons) make up the other 1% of carbon atoms and are virtually undetectable.
In PFAS analysis, labs typically add ¹³C-labeled PFAS to the samples prior to extraction.
For Example:
- Native PFOS = C₈F₁₇SO₃⁻
- ¹³C₄-PFOS = Same structure, but four carbons are ¹³C instead of ¹²C
Chemically, they behave identically during extraction and analysis, but the instrument can distinguish between the labelled and unlabeled version of the analyte because the masses are different.
The Colored Marbles Analogy
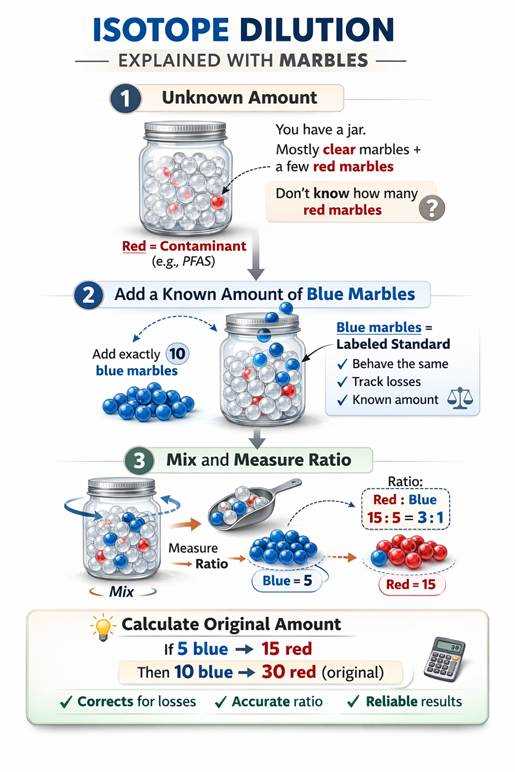
Imagine you have a big jar of clear marbles (your sample). Somewhere in that jar are a few red marbles but you dont know how many. Those red marbles represent PFAS in your sample. In order to determine how many red marbles are in the sample well use isotopic dilution.
1. Add a Known Number of Blue Marbles
You add exactly 10 blue marbles to the jar. These blue marbles represent the labeled isotope standard (¹³C-labeled PFOS). You know how many blue marbles you added.
2. Shake and Mix
Now everything is mixed together. The red marbles (native PFAS) and the blue marbles (labeled PFAS) behave exactly the same in the jar. This is the key idea: The labeled compound acts just like the native compound.
3. Scoop and Count the Ratio
You scoop out a portion and count:
- 5 blue marbles
- 15 red marbles
You know you added 10 blue marbles total. If half of your blue marbles show up in the scoop, then about half of the red marbles did too. Using the ratio of red to blue, you calculate the total number of red marbles in the jar.
How Isotopic Dilution Is Performed in the Lab
1. Spike the Sample with the Labeled PFAS Standard
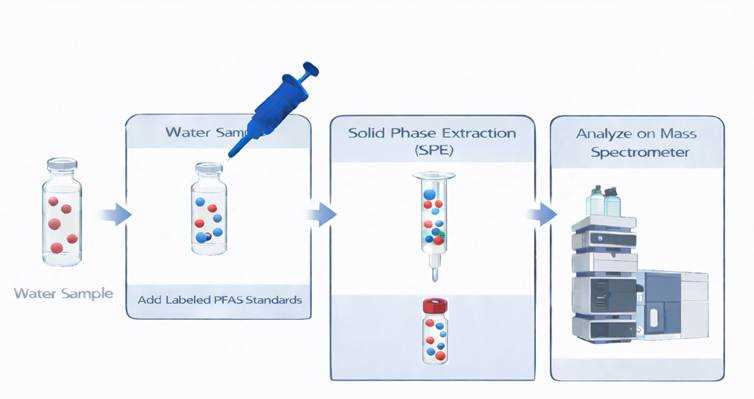
A known amount of ¹³C-labeled PFAS is added to the environmental sample before the extraction. This is critical — the isotope must experience every step the native PFAS does.
2. Extract the Sample by Solid Phase Extraction (SPE)
Both native PFAS and labeled PFAS:
- Load onto the SPE cartridge
- Wash
- Elute together
If recovery is 70%, both compounds lose 30% equally. Thats the power of the method.
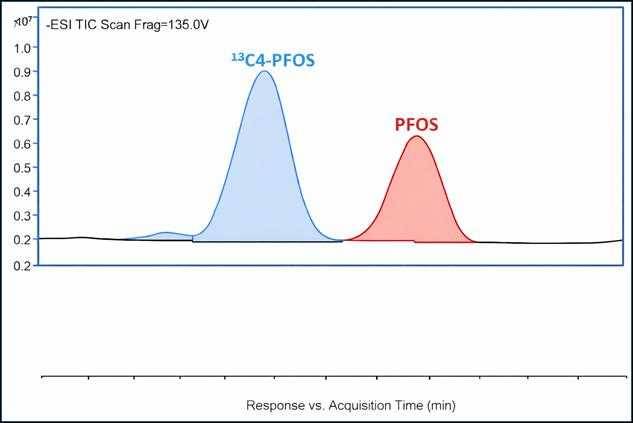
3. Analyze by LC-MS/MS
Analysis is typically performed using triple quadrupole LC-MS/MS. Since the native and isotope have different molecular weights, they can be distinguished from each other in the chromatogram.
🧠 Back to the Marble Analogy - Why Isotopic Dilution Works
- You know how many blue marbles you added.
- Blue and red marbles behave the same.
- Any losses affect both equally.
- The ratio stays accurate.
🧪 Relating the Marble Analogy to PFAS Analysis in the lab
- Red marbles = native PFOS in a water sample
- Blue marbles = ¹³C₄-PFOS internal standard
- Jar = your environmental sample
- Scooping = extraction + LC-MS/MS analysis
- Counting = measuring peak area ratios
Instead of counting marbles, the mass spectrometer measures the ratio of native PFOS signal to labeled PFOS signal.
|
Response Ratio =
Native PFAS Peak Area
|
Because both compounds behave identically, the ratio lets you calculate the true concentration, even if some material was lost during extraction.
Why Isotopic Dilution Is Superior to External Calibration
Without isotopic dilution:
- Losses during extraction cause under-reporting
- Matrix suppression causes inconsistent bias
- Results vary between instruments and analysts
With isotopic dilution:
- ✔ Recovery correction is built in
- ✔ Matrix effects are corrected
- ✔ Precision improves
- ✔ Data defensibility increases
- ✔ Regulatory compliance is achieved
For PFAS litigation and regulatory reporting, this level of defensibility is essential.
The Bottom Line
For PFAS analysis, isotopic dilution is not just a technique — it is the foundation of accurate, defensible environmental measurement.
If you're working as a consultant, regulatory testing, or litigation support, isotopic dilution isnt optional — its essential.
ALS has decades of expertise in PFAS analysis and isotope dilution workflows across challenging matrices.
If you're evaluating method options, data quality requirements, or turnaround/cost tradeoffs, contact ALS to discuss your project needs or request a quote — well help you select the most defensible approach for your samples and reporting goals.



























































